The order Phasmatodea contains more than 3,000 extant species of insect found throughout the world, especially the warmer zones. These herbivorous (mostly arboreal) insects are most well-known for their crypsis, or camouflage, where their colour, shape and behaviour enable them to masquerade as twigs or leaves (hence their common names of stick and leaf insects).
Though there is still uncertainty about a definitive phylogeny, phasmids are considered one of 11 orthopteroid insect orders within the assemblage known as Polyneoptera. Phasmatodea are found alongside the modern orders: Blattodea (cockroaches), Dermaptera (earwigs), Embioptera (web-spinners), Grylloblattodea (ice crawlers), Mantodea (praying mantises), Mantophasmatodea (heel-walkers) and, of course, Orthoptera (crickets, grasshoppers, and katydids). According to phylogenomic analyses of nucleotide and amino acid sequences, some of these Polyneopteran lineages are thought to have emerged ~302 million years ago, with phasmids evolving after the Permian mass extinction.1
Fossil Evidence
Fossil evidence of phasmids is, however, extremely rare. Specimens have been recovered in amber, most notably representatives of Euphasmatodea and Timematodea, with the oldest well-documented fossils being found in Cretaceous Burmese amber. Recent discoveries of the oldest-known fossilised leaf mimics (Phylliinae) from Messel, Germany in 2006 of Eophyllium messelensis date this foliacious mimicry to the Eocene.2
Although the fossil evidence is patchy, it is thought that traits relating to morphological plant masquerade within Phasmatodea first developed with stick mimicry in the Permian, followed by leaf mimesis developing in the Eocene when angiosperms largely replaced conifers as dominant trees.2

Crypsis, Camouflage, and Masquerade
In order to avoid visual detection by predatory mammals, birds, reptiles and other invertebrates, many insects evolved morphological characteristics that enabled them to blend in to their surrounding environment. Specifically in the case of phasmids, these evolutionary adaptations have been very closely linked to the insects’ host and food plants leading to a coupling of ecological and evolutionary dynamics.4,5,6
Cryptic colouration, elongation of the body and legs, or, alternatively, broadening and flattening of the body to resemble leaves are all forms of masquerade adopted by phasmids to avoid detection by predators. This has led some researchers to conclude that predation may be an important driver of speciation in this order. Successful adaptation, through camouflage, may therefore lead to divergence in adaptive radiation. 4,5,6
The basal-most extant recorded clade of Phasmatodea is the sub-order Timematodea, within which is found the genus Timema whose species are found throughout southwestern North America on a variety of host-plant species.7 Experimental studies have been conducted into the influence of ecological factors with regards adaptive radiation in Timema cristinae with particular emphasis on host plants and cryptic colouration.4,5,6,7

It was found that two distinct T. cristinae morphs had developed on two morphologically dissimilar plant species distributed in parapatric mosaics. The first plant, Adenostoma fasciculatum, has needle-like leaves, while the other, Caenothos spinosus, has broad, ovate leaves. Each T. cristinae morph (or ecotype) was found to be more cryptic on one of the two plant species depending on whether they displayed a heritable white dorsal stripe or not. One of the experiments found that bird predation significantly lowered numbers of T. cristinae that were maladapted to the host plant.4 Further studies concluded that through predator pressure, partial (but incomplete) ecological speciation has occurred in T. cristinae as the morphs still successfully interbred.5,6,7
This partial speciation may, however, form only one dimension of selective pressures that constitute adaptive radiation events. It was also shown that by comparing different Timema species that share the same host plant, that sexual isolation was not as marked as with between species on different plants (when compared with T. podura and T. chumash)7 lending credence to the notion of ecological speciation. It is therefore apparent that predators apply selective pressure leading to morphological crypsis and divergence, but that this does not necessarily directly lead to speciation, but is more likely an intermediate stage in adaptive radiation.
Flying, Jumping, and Holding Still
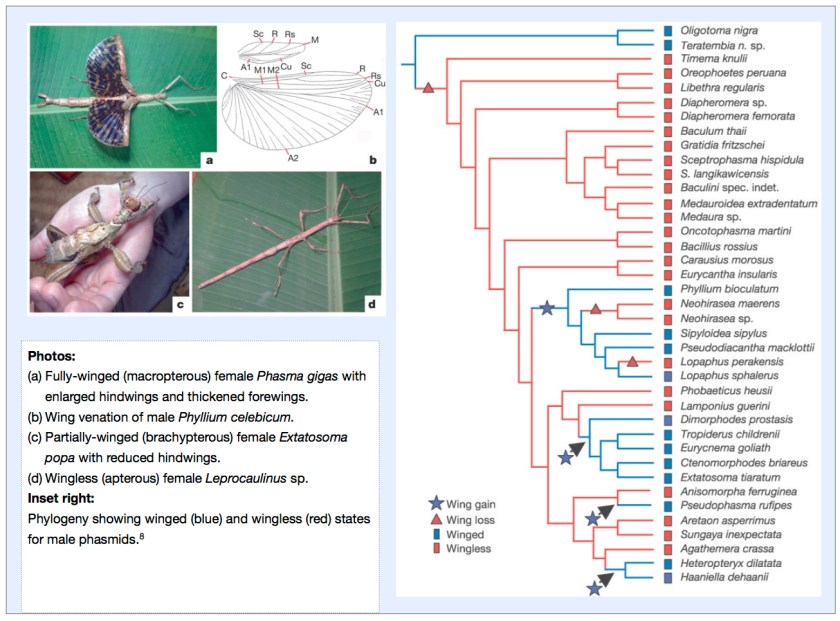
In the case of the Phasmatodea, it has been discovered that diversification occurred in a wingless state and that wings were subsequently derived on a number of occasions.
Of the 3,000 species of phasmids, only 40% are fully winged, while the remainder are partially winged or entirely wingless. While being fully winged conveys advantages of dispersal, escape and finding resources, it has been claimed that increased female fecundity and crypsis may have served as a selective advantage in early phasmid evolution in a shift to winglessness. Apart from flight, wings and partial wings can also be used in threat or startle responses to deter would-be predators. By examining DNA sequence data and applying parsimony optimisation it was found that the ancestral condition of Phasmatodea is wingless.8
It was also found that certain phasmid lineages had “re-evolved” wings prompting suggestions that this reacquisition may confer adaptive advantages of being both winged and wingless as conditions necessitate over ecological time leading to further speciation.8
Timema chumash is unusual in phasmids in that it has been found to jump away from potential threats. Although it can only jump relatively short distances by extending the hind tibia, it can reach take-off velocities comparable to some larger European flea beetles. The leg positions and hind-leg length of T. chumash contrast with the morphology of other stick insects; its legs emerge ventrally from the thorax and its hind legs are proportionately longer than those of other phasmids. As T. chumash is wingless, it jumping is suggestive that it would enable a rapid fall from the plant it was perching on taking it out of the visual field of predators and providing it with another opportunity to camouflage itself nearby.9 These morphological and behavioural traits may present opportunities for further adaptive evolution.

A well-documented behaviour in many phasmids is that of catalepsy whereby the insect is able to remain motionless or produce extremely slow movement as a form of twig or leaf mimesis to aid with predator evasion. The mechanism by which this is achieved, is via the high gain of the femur-tibia joint control system,10 and has been recorded in fossil specimens.2 A key difference between phasmids and other orthopteroids is this significant coevolution of the mimetic body shape with catalepsy.10
Parthenogenesis, Hybridogenesis and Androgenesis
Phasmids experience a wide array of reproductive modes with about 10% of the phasmid taxa being parthenogenetic and producing all-female offspring (thelytoky).11 Although parthenogenesis reduces genetic variability, it does not wholly suppress it. Furthermore autopolyploids and allopolyploids can take advantage of higher mutational rates to increase heterozygosity. Androgenesis is also common and has been proposed as a likely pathway to cladogenesis in the genus Clonopsis11 and has already been recorded in Pijnackeria where tetraploid hybrids lacking maternal genes, but keeping the maternal mitochondrial DNA, speciated.12 The discovery of interracial and interspecific hybridogenesis in the genus Bacillus added further weight to the notion of maintaining (or even increasing) genetic diversity within phasmid lineages and creating opportunities for further speciation. 12
No one reproductive mechanism is exclusively used, so that complete reversion from thelytoky to amphimixis is possible. These “tangled interactions” allow for genetic diversity to persist within and between populations. When considered as part of a series of repeated and complex reproductive strategies including sexual reproduction, parthenogenesis, androgenesis and hybridogenesis, it must be concluded that evolutionary pathways for phasmids are far from dead-ends.11, 12
Following divergence from other orthopteroids, phasmids took advantage of the new food sources and flourished following the angiosperm revolution and have continued to adapt in relation to predatory pressures, host-plant availability, behaviours, and complex reproductive strategies. Clearly, apart from the opportunities presented in the Eocene for cladogenesis and speciation, there continues to be further evolutionary opportunities relating specifically to morphology and sexual isolation in adaptive radiation of phasmids.

References:
1. Misof, B. et al. (2014), Phylogenomics resolves the timing and pattern of insect evolution. Science. 346 (610), 763-767.
2. Wedmann, S., Bradler, S., and Rust, J. (2006), The first fossil leaf insect: 47 million years of specialized cryptic morphology and behavior. Proceedings of the National Academy of Sciences. 104 (2), 565-569.
3. Wang, M., Be´thou, O., Bradler, S., Jacques, FMB., Cui, Y., and Ren, D. (2014), Under Cover at Pre-Angiosperm Times: A Cloaked Phasmatodean Insect from the Early Cretaceous Jehol Biota. PLoS One, 9 (3), e91290
4. Farkas, TE., Mononen, T., Comeault, AA., Hanski, I. and Nosil, P. (2013) Evolution of Camouflage Drives Rapid Ecological Change in an Insect Community. Current Biology. 23, 1835-1843.
5. Nosil, P., Crespi, BJ., and Sandoval, CP. (2002) Host-plant adaptation drives the parallel evolution of reproductive isolation. Nature. 417, 440-443.
6. Nosil, P. and Crespi, BJ. (2006) Experimental evidence that predation promotes divergence in adaptive radiation. Proceedings of the National Academy of Sciences. 103 (24), 9090-9095.
7. Nosil, P. and Sandoval, CP. (2008) Ecological Niche Dimensionality and the Evolutionary Diversification of Stick Insects. PLoS One. 3(4), e1907
8. Whiting, MF., Bradler, S. and Maxwell, T. (2003) Loss and recovery of wings in stick insects. Nature. 421, 264-267.
9. Burrows, M. (2008) Jumping in a wingless stick insect, Timema chumash (Phasmatodea, Timematodea, Timematidae). The Journal of Experimental Biology. 211, 1021-1028.
10. Wolf, H., Bässler, U., Spieß, R. and Kittman, R. (2001) The femur–tibia control system in a proscopiid (Caelifera, Orthoptera): a test for assumptions on the functional basis and evolution of twig mimesis in stick insects. The Journal of Experimental Biology. 204, 3815-3822.
11. Scali, V. (2009) Stick insects: parthenogenesis, polyploidy and beyond. In: Life and Time: The Evolution of Life and its History. Cleup, Padova. 171-192